
New titanium alloys of the beta-type or metallic glasses (amorphous alloys) are promising new materials which can meet the high demands on bone implants in orthopedics, trauma surgery or dentistry regarding mechanical compatibility, corrosion resistance and biocompatibility. Innovative non-equilibrium processing from metal casting to additive manufacturing (SLM, LPBF, 3D print) pave new paths for patient-specific implant designs. We investigate relations between the composition- and process-conditioned microstructure and morphology of those new alloys and their resulting mechanical properties as well as the corrosion behavior in physiological solutions. A main focus of our research is the generation of tailored surface states of these novel alloys to achieve highest corrosion resistance and to allow for optimal interaction with bone cells/tissues. A new key aspect are functional surfaces with antibacterial properties to reduce the risks of implant-related inflammation reactions and infections.
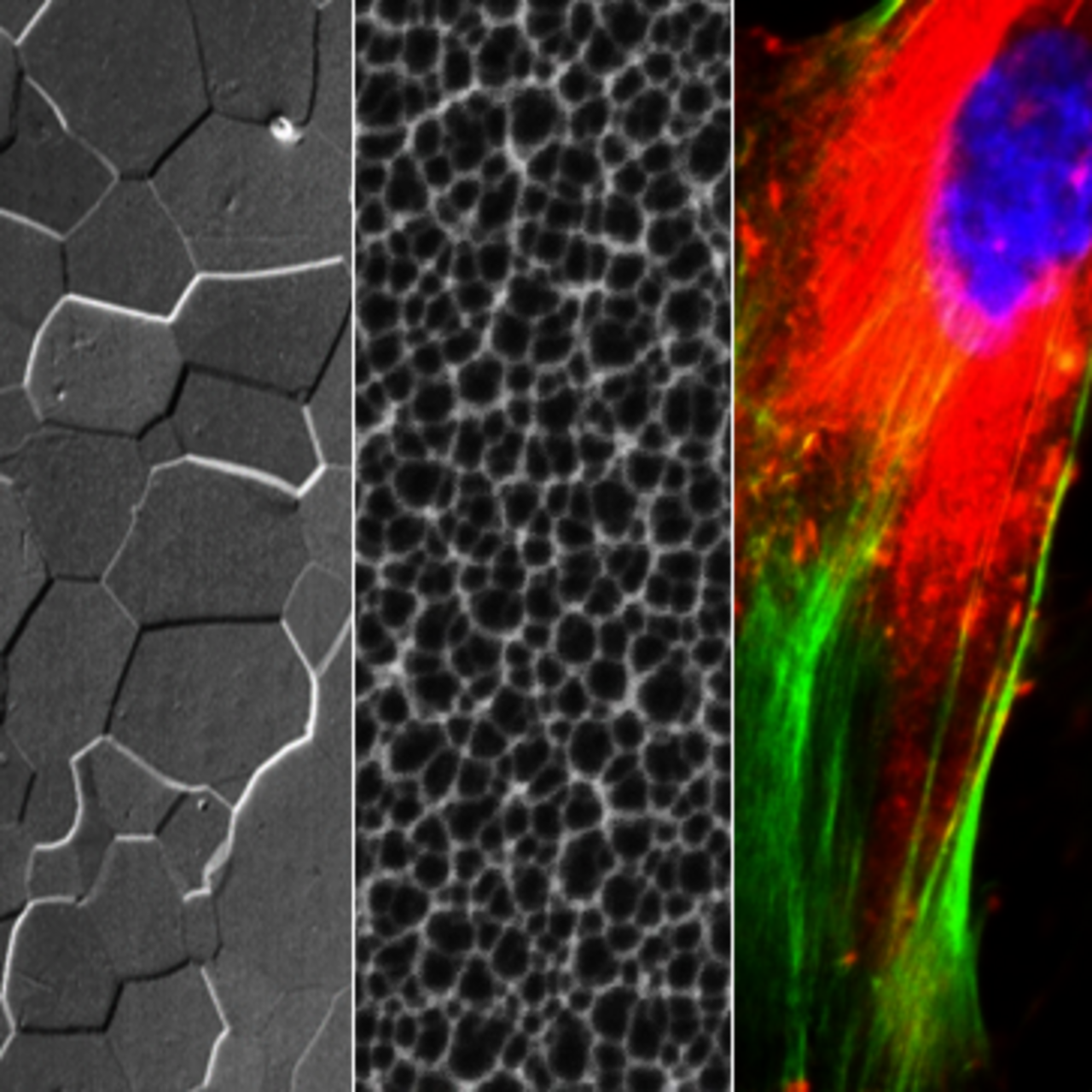
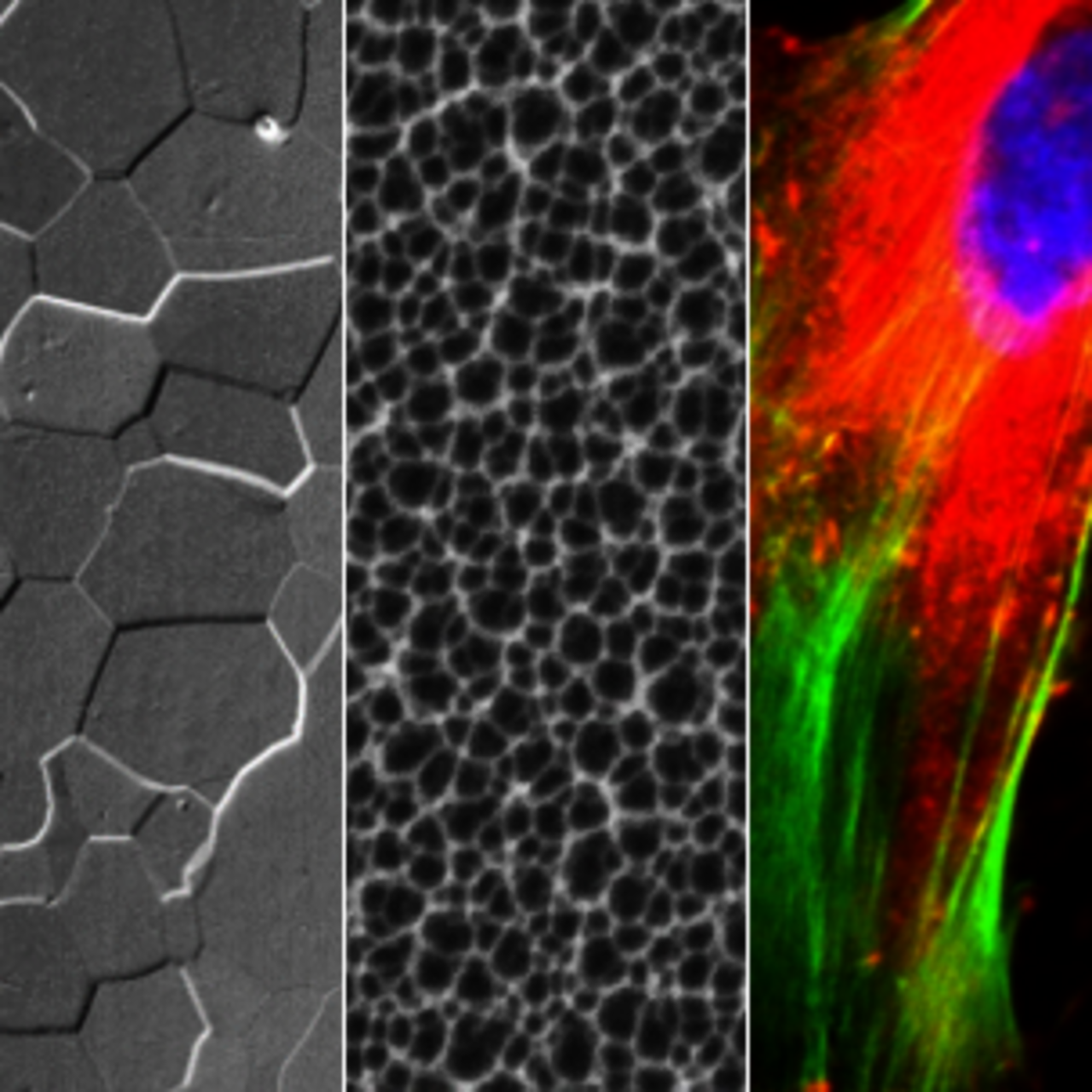
We work in close cooperation with IFW groups at IMC, IMW, IFF.
Patient-specific designs and new metallic materials with bone-adapted mechanical properties and biocompatible compositions are mandatory requirements for advanced bone implants. Additive manufacturing technologies such as Laser Powder Bed Fusion (LPBF, PBF-LB/M) enable the near-net shape production of complex implant designs. As a non-equilibrium technique, LPBF can also generate defined microstructural states, thereby yielding unique mechanical performance. This is specifically promising for beta-type (β) Ti-based alloys which combine the benefits of low elastic moduli and non-toxic alloy constituents.
On the example of the metastable Ti-42Nb alloy, we have demonstrated how LPBF processing and post heat treatments can control the mechanical properties. By using a non-standard top hat laser source, bulk specimen in a single-phase beta-phase state and with a strong <001> texture parallel to the building direction (BD) were fabricated. Tensile tests along selected loading directions revealed the strong mechanical anisotropy with the lowest Young’s modulus of only 44 GPa parallel to BD. This is in accordance with the anisotropy of mechanical properties of long bone and can be exploited for load-bearing implants.
Suitable post heat treatment strategies for LPBF-produced Ti-42Nb were explored to achieve substantial materials strengthening effects. For example, heat treatments within the α+β phase range at 723 K for different durations led to increasing fractions of the metastable orthorhombic α’’ iso phase, acting as an intermediate to the stable α phase. Micromechanical analysis was used for assessing the impact of time-dependent microstructural changes on the evolution of reduced modulus (Er) and hardness (H) values. Tensile tests demonstrated a significant strength enhancements through treatments up to 108 ks compared to the as-built state, thereby reaching the performance level of clinical benchmark materials such as Ti-6Al-4V.
The gained knowledge on those strategies for combined LPBF and post heat treatments were successfully employed to achieve enhanced mechanical and corrosion performance of additively manufactured near-beta Ti-13Nb-13Zr via microstructural design.
Further, LPBF (PBF-LB/M) is a powerful technology for the fabrication of metallic scaffolds with low structural stiffness. New concepts refer to lattices which are designed on basis of triple periodic minimal surfaces (TPMS). Those TPMS lattices such as the gyroid type have biomimetic morphologies and exhibit in comparison to classical lattices improved biological, mechanical and fluiddynamic properties. In collaboration with a team at the TU Dresden we have developed a numerical-experimental approach for the optimal design of those TPMS lattices for bone implants and their fabrication with LPBF using Ti-42Nb alloy powder. A key activity was the evaluation of surface-related imperfections and shape deviations of real lattices and their impact on the mechanical performance. In consequence, numerical solutions for lattice shape optimization and chemical post treatments for reducing surface imperfections were successfully developed.
Günther, F., Pilz, S., Gebert, A., Zimmermann, M. et al., Defect-dependent fatigue properties of ß Ti-42Nb processed by laser powder bed fusion, Mater. Today Comm. 43 (2025) 111733.
doi.org/10.1016/j.mtcomm.2025.111733
Pilz, S., Bönisch, M., Gebert, A. et al., Tailoring microstructure and mechanical properties of an LPBF-processed beta Ti-Nb alloy through post-heat treatments, Mater. Des. 239 (2024) 112799.
doi.org/10.1016/j.matdes.2024.112799
Pilz, S., Hariharan, A., Gebert, A. et al., Influence of isothermal omega precipitation aging on deformation mechanisms and mechanical properties of a β-type Ti-Nb alloy, J. Alloys Compds. 930 (2023) 167309.
doi.org/10.1016/j.jallcom.2022.167309
Pilz, S., Günther, F., Zimmermann, M., Gebert, A. et al., Controlling the Young’s modulus of a ß-type Ti-Nb alloy via strong texturing by LPBF, Mater. Design 216, (2022) 110516.
doi.org/10.1016/j.matdes.2022.110516
Hariharan, A., Goldberg, P., Pilz, S., Schell, F., Gebert, A. et al., Designing the microstructural constituents of an additively manufactured near β Ti alloy for an enhanced mechanical and corrosion response, Mater. Design 217 (2022) 110618.
doi.org/10.1016/j.matdes.2022.110618
Günther, F., Pilz, S., Hirsch, F., Gebert, A., Zimmermann, M. et al., Shape optimization of additively manufactured lattices based on triply periodic minimal surfaces, Add. Manuf. 73 (2023) 103659.
doi.org/10.1016/j.addma.2023.103659
Günther, F., Pilz, S., Hirsch, F., Gebert, A., Zimmermann, M. et al., Experimental and numerical characterization of imperfect additively manufactured lattices based on triply periodic minimal surfaces, Mater. Des. 233 (2023) 112197.
doi.org/10.1016/j.matdes.2023.112197
Günther, F., Pilz, S., Hirsch, F., Gebert, A., Zimmermann, M. et al., Structure-property relationships of imperfect additively manufactured lattices based on triply periodic minimal surfaces, Mater. Des. (2022) 111036.
doi.org/10.1016/j.matdes.2022.111036
Project: EU EFRE SAB OsteoLas (grant no. 100382988 / 10382989), DFG (grant no. 419952352)

Beta-type (β) Ti-based alloys are promising candidates for bone implant applications due to their low Young’s modulus values providing low implant stiffness, good workability and ductility, and outstanding biocompatibility. However, for specific load-bearing implants the wear resistance in biological environment is a decisive aspect.
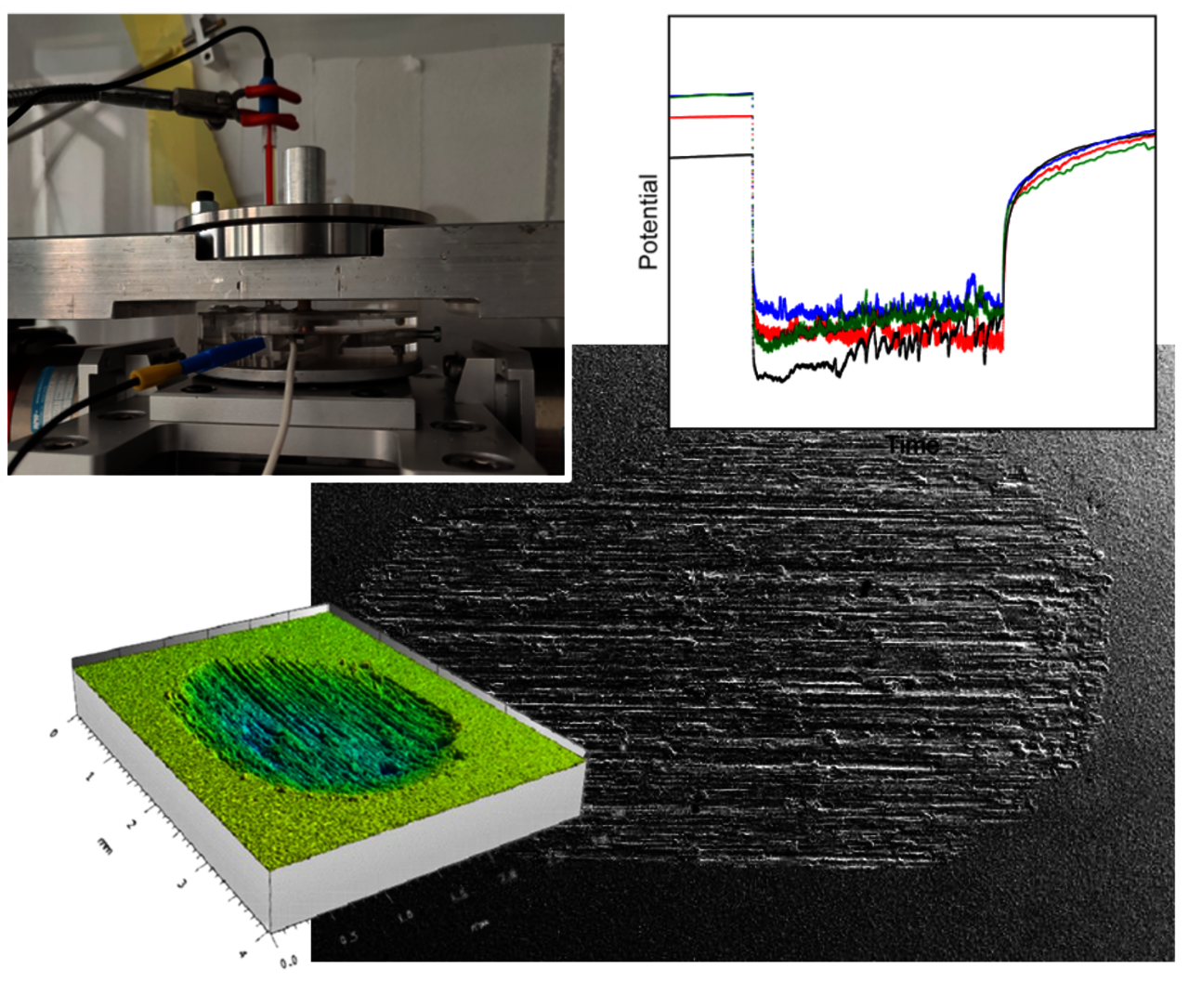
In collaboration with a team at the University Lyon, INSA de Lyon, we have investigated mechanically-assisted corrosion phenomena, such as tribocorrosion, for the additively manufactured beta-type Ti-42Nb alloy. Laser Powder Bed Fusion (LPBF, PBF-LB/M) yields single-phase bulk specimen with unique refined microstructural states compared to cast and solution-treated alloy states. In consequence, the LPBF-built alloy states exhibit a higher tribocorrosion resistance in a synthetic body fluid. The detected volume losses are mainly due to mechanical wear owing to the strong re-/passivation ability of those beta-type alloys. Differences in volume losses for various materials states correlate with their respective microhardness.
Post heat treatments of LPBF-fabricated Ti-42Nb specimen resulting in the precipitation of ω or α’’ iso phases do not change the passivity but affect the tribocorrosion resistance.
Akman, A., Hariharan, A., Pilz, S., Zimmermann, M., Gebert, A., Influence of isothermal post-treatment on tribocorrosion behaviour of laser powder bed fusion-processed -type Ti-Nb alloy, Today Commun. 50 (2026) 114222.
doi.org/10.1016/j.mtcomm.2025.114222
Akman, A., Alberta, L.A., Gebert, A. et al., Tribocorrosion behaviour of additively manufactured β-type Ti–Nb alloy for implant applications, J. Mater. Res. Technol. 31 (2024) 1419.
doi.org/10.1016/j.jmrt.2024.06.172
Project: EU-ITN BIOREMIA (grant no. 861046), DFG (grant no. 419952351)
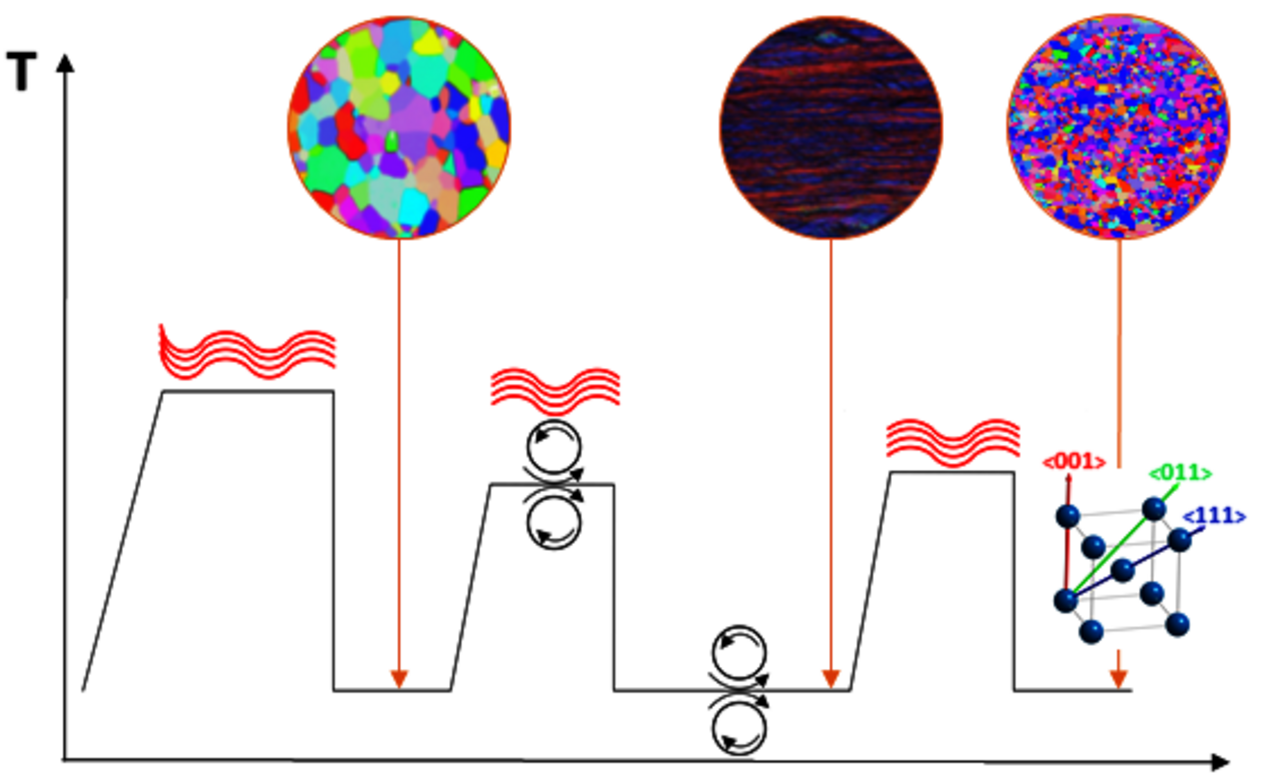
Beta-type (β) Ti-(40-45)Nb alloys are promising materials for load-bearing bone implants in orthopaedics and trauma surgery. They exhibit very low Young’s moduli of ~60 GPa yielding a very low implant stiffness which contributes to the reduction of stress shielding effects.
For achieving optimum mechanical biofunctionality, different hardening strategies based on complex warm and cold rolling treatments in combination with annealing were developed. Most effective are routes comprise grain boundary hardening with preservation of the β-phase and further work hardening or precipitation hardening. Low temperature annealing (300°C) of recrystallized Ti-40Nb samples caused nanoscale ω-precipitations. Those stabilize the β-phase against stress-induced martensite formation and significantly increase the tensile strength to values >900 MPa which is comparable to values for Ti-6Al-4V. Further, the fatigue resistance is markedly increased.
Alberta, L.A., Gebert, A. et al., Effect of aging treatments on mechanical, chemical, and antibacterial properties of a β-type Ti–Nb–Cu alloy for implant applications, J. Mater. Res. Technol. 36 (2025) 1049.
Pilz, S. et al., Influence of isothermal omega precipitation aging on deformation mechanisms and mechanical properties of a β-type Ti-Nb alloy, J. Alloy. Compd. 930 (2023) 167309.
Pilz, S. et al., Thermomechanical processing of In-containing ß-type Ti-Nb alloys, J. Mech. Behav. Biomed. Mater. 79 (2018) 283.
Helth, A. et al., Effect of thermomechanical processing on the mechanical biofunctionality of a low modulus Ti-40Nb alloy, J. Mech. Behav. Biomed. Mater. 65 (2017) 137.
Calin, M. et al., Elastic softening of Beta-type Ti-Nb alloys by indium (In) additions , J. Mech. Behav. Biomed. Mater. 39 (2014) 162.
Project: DFG SFB Transregio 79
The application of powder metallurgy for β-Ti-Nb alloys enables new developments towards a variety of near-net shape techniques as well as tailored implants with an porosity for permanent bone implants. The introduction of an open porosity is advantageous for bone ingrowth and allows significant reduction of the implant stiffness.
Ti-(40-45m.%)Nb samples were successfully prepared by hot pressing or sintering of mechanically alloyed or gas-atomized β-TiNb powder without/with space holder phase. Due to their refined microstructure, fully dense samples show a significantly higher yield strength >900 MPa than conventionally casted β-TiNb-alloys. Scaffolds with 30-70% porosity were produced with NaCl as space holder, with very low Young’s modulus (10-60 GPa) but only limited yield strength. The empirical determination of the relationship between porosity and Young’s modulus was further assessed by finite element analysis.
Schmidt, R. et al., Powder metallurgical processing of low modulus beta-type Ti-45Nb to bulk and macro-porous compacts, Powder Technol. 322 (2017) 393.
Zhuravleva, K. et al., Phase transformations in ball-milled Ti-40Nb and Ti-45Nb powders upon quenching from the beta-phase region, Powder Technol. 253 (2014) 166.
Zhuravleva, K. et al., Determination of the Young’s modulus of porous beta-type Ti-40Nb by finite element analysis, Mater. Des. 64 (2014) 1.
Zhuravleva, K. et al., Mechanical alloying of beta-Type Ti-Nb for biomedical applications, Adv. Eng. Mater. 4 (2013) 262.
Project: DFG SFB Transregio 79

Alternative methods to the conventional powder production process were investigated to synthesis ultrafine-grained Ti and TiNb-powders for the production of alloys with a high strength to Young’s modulus ratio. Starting from pure Ti and Nb powders, β-type Ti-40Nb with a submicron grain size was produced by reactive milling in hydrogen atmosphere and subsequent thermal desorption treatment. Furthermore, a calciothermic synthesis route was developed to produce hydrogenated Ti and Ti–Nb powders directly from oxides. Fine grained powders with a particle size well below 45 µm and a rounded shape were achieved. These hydrogenated powders are suitable for the convectional press and sintering technique or MIM.
Lindemann, I. et al., Calciothermic Synthesis of Very Fine, Hydrogenated Ti and Ti–Nb Powder for Biomedical Applications, Adv. Eng. Mater. 3 (2019) 1901210/1-7
Lindemann, I. et al., Ultrafine-grained Ti-40Nb prepared by reactive milling of the elements in hydrogen, J. Alloys Compd. 729 (2017) 1244
Lindemann, I. et al., Synthesis of spherical nanocrystalline titanium hydride powder via calciothermic low temperature reduction, Scr. Mater. 130 (2017) 256
Projects: DFG

Owing to their chemical composition and their single-phase nature, beta Ti-Nb alloys are highly corrosion-resistant materials. In body fluids they exhibit extremely low metal release rates, excellent passivity and no indication for local corrosion. This gives rise to a very high biological compatibility. However, alloying additions or second phases which can further improve their mechanical biofunctionality or generate antibacterial effects may have an impact on their corrosion stability and this has to be assessed. As an example, it was shown that small indium (In) additions to Ti-40Nb reduce the Young’s modulus of the alloy. In simulated body fluids the release of ionic metal species from (Ti-40Nb)-In is insignificant. Indium is homogeneously incorporated in the beta-phase, surface passivation with Ti- and Nb-oxides occurs with traces of In-oxides in the passive films on as-ground states as well as on alloy surfaces, which were chemically etched for nanoroughening. In vitro studies (TU Dresden, U. Hempel) revealed a better hBMSC activity on the beta-type alloys in comparison to CP-Ti. This is mainly due to their high Nb content, which alters the passive layer composition. Modified cell adhesion and increased TNAP activity were observed when Indium is alloyed to Ti-40Nb.
A decisive aspect for bone implants made of beta Ti-Nb alloys is the creation of bioactive surface states with suitable topography, roughness, composition, surface energy etc. that enable optimum bone tissue ingrowth. On the other hand, advanced implants should exhibit appropriate antibacterial properties. We develop strategies and methods for nano- and micro-structuring of beta-type alloy surfaces by chemical and electrochemical treatements in order to achieve those optimum biofunctionalities.
The extremely high corrosion resistance of Ti-Nb alloys restricts surface treatments, e.g. standard chemical etchings, which are established for conventional implant materials. Nanoroughening of Ti-Nb surfaces together with enhanced passive film growth was obtained by exposure to oxidative acid solution. Those surface states were found to support the apatite-forming ability and to stimulate the response of human mesenchymal stromal cells (TU Dresden, U. Hempel). Anodization treatments under defined conditions can yield oxide nanotubes, microporous oxide coatings or compact oxide films whereby the high Nb content of the alloys has significant impact on the growth processes and on the specific oxide layer properties. Nb species are involved in the oxidation processes which causes significantly enhanced layer thickness growth, morphological changes and formation of mixed oxides (Ti,Nb)O2. Another approach comprises coatings of Ti-Nb surfaces with bioactive, osteoconductive Ca-phosphate compounds. The applicability of electrodeposition processes for the generation of adhesive hydroxyapatite coatings with different morphologies was demonstrated and the underlying electro-reduction and chemical precipitation steps were analyzed. In particular with galvanostatic pulse deposition the formation of nanocrystalline layers can be controlled. By variation of the solution composition the incorporation of Sr-species in the hydroxyapatite structure, which are important for osteoporosis treatment, was obtained. The Sr-content was found to determine layer morphology and thickness. In vitro studies with hBMSC (TU Dresden. A. Lode) revealed that the released Sr-ions led to a significantly enhanced cell proliferation and osteogenic differentiation and that the Sr-HAp surface supported cell adhesion indicating its excellent cytocompatibility. This coating method is also applicable to microporous Ti-Nb specimen produced by powder technological techniques.
A. Akman, A. Gebert et al., Effect of minor gallium addition on corrosion, passivity, and antibacterial behaviour of novel β-type Ti–Nb alloys, J. Mater. Res. Technol. 25 (2023) 4110.
R. Schmidt, A. Gebert et al., Electrodeposition of Sr-substituted hydroxyapatite on low modulus beta-type Ti-45Nb and effect on in vitro Sr release and cell response, Mater. Sci. Eng. C 108 (2020) 110425.
J. Vishnu, M. Calin, …, G. Manivasagam et al., Superhydrophilic nanostructured surfaces of beta Ti-29Nb alloy for cardiovascular stent applications, Surf. Coat. Technol. 396 (2020) 125965.
S. Pilz, A. Gebert et al., Metal release and cell biological compatibility of beta-type Ti-40Nb containing indium J. Biomed. Mater Res. B 106, (2018) 1686.
A. Gebert, …, M. Rohnke, J. Janek et al., Oxidation treatments of beta-type Ti-40Nb for biomedical use, Surf. Coat. Technol. 302 (2016) 88.
P.F. Gostin at al., Surface treatment, corrosion behaviour and apatite-forming ability of Ti-45Nb implant alloy, J. Biomed. Mater. Res. B 101 (2013) 269.
Project: DFG SFB Transregio 79, EU ITN BIOREMIA

Metallic glasses are known to combine high strength and hardness with comparatively low Young‘s modulus values. This has raised the interest to evaluate in particular Ti-based glasses as possible biomaterials. However, alloy design demands a compromise between the achievement of high glass-forming ability and the use of non-toxic constituents. Alloys of the Ti-Zr-Si(-Nb) system can be rapidly quenched to glassy or glass-matrix (nanocomposite) states. Mechanical testing predicts high wear resistance. Corrosion tests in Ringer’s solution revealed very low corrosion rates similar to those of beta-type Ti-Nb and stable anodic passivity. Nb addition improves the glass-forming ability and the mechanical properties and also supports a high pitting resistance even at extreme anodic polarization. Thermal treatments are possible in a defined temperature-time window to preserve alloy’s microstructure and yield oxide films with characteristic morphologies. The apatite-forming ability was proven. In collaboration with Univ. Pardubice the generation of self-organized double-wall oxide nanotube layers was demonstrated. These properties recommend these new alloys for application as coating materials in the biomedical field.
Shtefan, V., Fernández-Navas, N., Nemec, T., Gebert, A. et al., Copper Nanocrystallization in Anodic Oxide Films of Ti-Cu-based Bulk Metallic Glass and Its Effect on Corrosion Resistance and Cytocompatibility, Advanced Engineering Materials (2026) e70774.
Fernández-Navas, N., Shtefan, V., Voss, A., Goldberg, P., Gebert, A. et al., Improved biocompatibility of Ti-based bulk metallic glasses by electrochemical formation of nanoporous oxide layers, Biomaterials Advances 177 (2025) 214378.
Shtefan, V., Fernández-Navas, N., Gebert, A. et al., Pitting Corrosion Mechanisms of Ti-Cu-(Pd-) Based Metallic Glasses in Simulated Physiological Solution, Corros. Sci. 251 (2025) 112913.
Fernández-Navas, N., Shtefan, V., Gebert, A. et al., Electrochemical Surface Nanostructuring of Ti47Cu38Fe2.5Zr7.5Sn2Si1Ag2 Metallic Glass for Improved Pitting Corrosion Resistance, Adv. Eng. Mater. 26 (2024) 2302206.
Fernández-Navas, N., Shtefan, V., Gebert, A. et al., Acid Treatments of Ti-Based Metallic Glasses for Improving Corrosion Resistance in Implant Applications, Metals 14 (2024) 241.
H. Sopha, D. Pohl, …, A. Gebert, J. Macak, Self-organized double-wall oxide nanotube layers on glass-forming Ti-Zr-Si(-Nb) alloys, Mater. Sci. Eng. C 70 (2017) 258.
S. Abdi, M. Bönisch et al., Thermal oxidation behavior of glass-forming Ti-Zr-(Nb)-Si alloys, J. Mater. Res. 31 (2016) 1264.
S. Abdi et al., Corrosion behavior and apatite-forming ability of glassy Ti75-xZr10NbxSi15 (x=0,15) alloys – potential materials for implant applications, J. Biomed. Mater. Res. Part B 104 (2016) 27.
S. Abdi et al., Effect of Nb adition on microstructure evolution and nanomechanical properties of a glass-forming Ti-Zr-Si alloy, Intermetallics 46 (2014) 156.
M. Calin at al., Designing biocompatible Ti-based metallic glasses for implant applications, Mater. Sci. Eng. C 33 (2013) 875.
Project: EU-ITN BIOTINET
In an effort to develop novel biocompatible glassy metallic materials with an appropriate combination of biomedical properties as well as MRI compatibility, we have adopted a new composition design strategy based on the high-entropy alloys approach by exploring the central region of multi-component alloy phase space of the Ti-Zr-Nb-Hf-Si system. The newly developed multi-principal element alloys have biocompatible compositions, with magnetic susceptibility tailored to the medical MRI environment. They exhibit high thermal stability and excellent corrosion stability in simulated body fluid. All alloys show a weak paramagnetic nature advantageous for reducing the MRI artefacts. In addition, they exhibit higher X-ray linear attenuation coefficients relevant for interventional X-ray-based medical imaging. This two-fold advantage (lower magnetic susceptibility and higher radiopacity) allows the materials to be more precisely visualized via biomedical imaging methods, which is especially important for miniaturised implants such as coronary stents or aneurysm clips.These results provide a guideline on how to design equiatomic multicomponent metallic glasses with unique combination of biomedical properties by utilizing HEA characteristics, and thus providing a strategy for bridging MGs and HEAs fields for the discovery of new alloys of scientific significance and practical benefit.
Nano- and micro-patterning of biomaterials is a rapidly evolving technology used in the engineering sciences to control cell behavior. The surface topography of the implant material plays an important role in enhancing the cell-material interaction. However, the practical applicability of nano-/micro-patterning strategies with metals is restricted due to intrinsic limits in processability of high-strength metals. Bulk metallic glasses (BMGs) constitute a class of materials ideally suited to surface patterning in view of biomedical applications. They exhibit a unique temperature-dependent mechanical behavior, which enables polymer-like processability at temperatures slightly higher than the specific glass transition temperature of BMGs. We studied the thermoplastic forming ability of Ti- and Zr-based BMGs with potential biomedical applications. The unique processability and intrinsic properties of this new class of amorphous alloys make them competitive with the conventional metallic biomaterials.
In a cooperation work with partners from VitriMetTech EU project (http://www.vitrimettech.unito.it/) the unique thermoplastic behavior of Ni-free Ti- and Zr-based bulk metallic glasses has been investigated by utilizing the dramatic softening of the amorphous structure in the super cooled liquid region.
B. Sarac et al., Micropatterning kinetics of different glass-forming systems investigated by thermoplastic net-shaping, Scr. Mater. 137 (2017) 127
S. Bera et al., Micro-patterning by thermoplastic forming of Ni-free Ti-based bulk metallic glasses, Mater. Des. 120 (2017) 204
Project: EU-ITN VitriMetTech

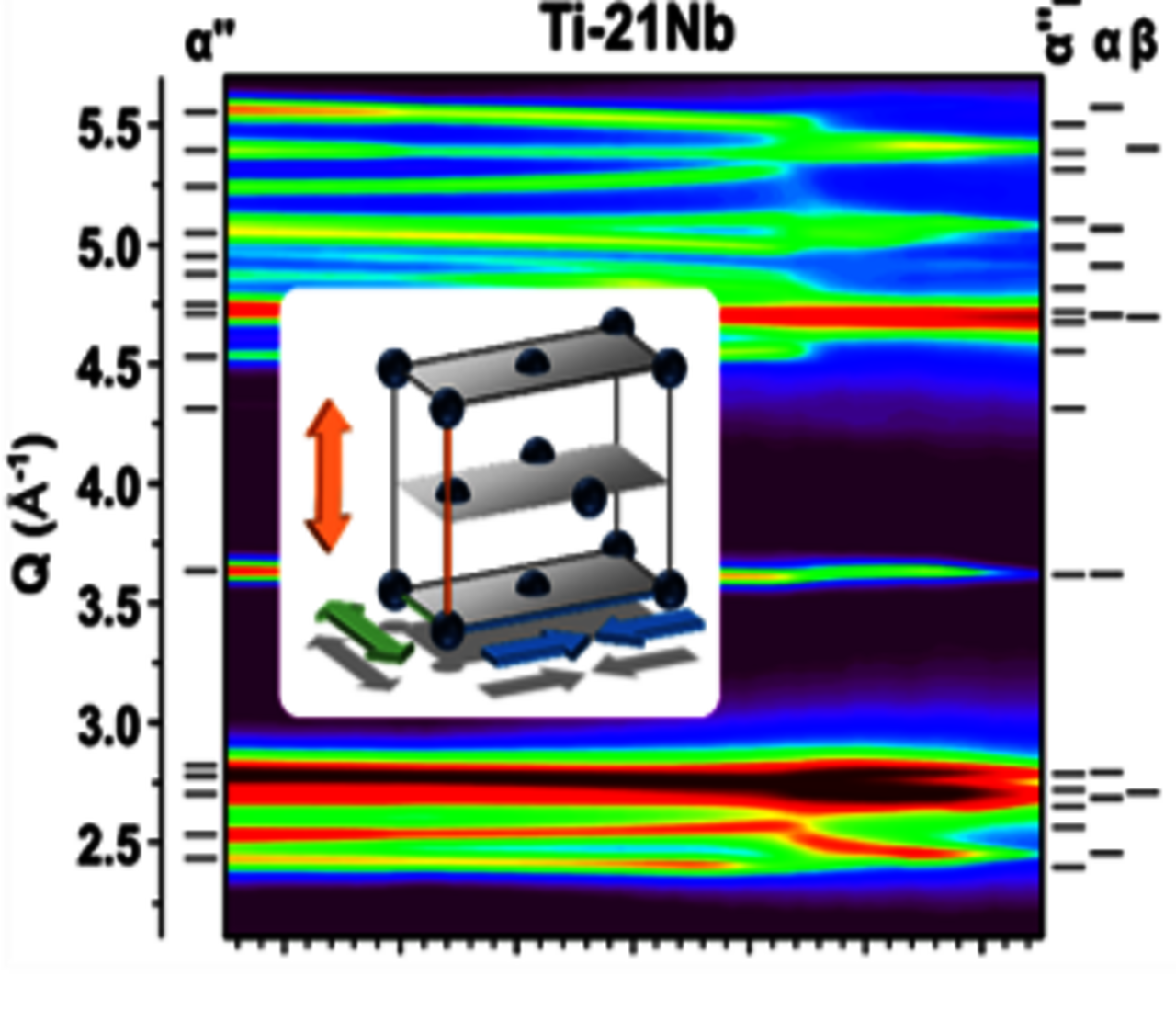
Ti-alloys represent the principal structural materials in both metallic biomaterials and aerospace design development. Key to optimizing their mechanical and functional behaviour is in-depth know-how of their phases and the complex interplay of diffusive vs. displacive phase transformations to permit the tailoring of intricate microstructures across a wide spectrum of configurations.
In particular the class of β-stabilized Ti-alloys represents highly promising multifunctional materials with desirable structural and functional properties for various biomedical and engineering applications. Due to the thermoelastic martensitic transformation of the body centred cubic (bcc) β-phase to orthorhombic martensite α″ this alloy family also demonstrates shape memory (SM) behavior and superelasticity (SE). Up to the present day, these features have been stirring ever increasing interest.
Since the discovery of SM in Ti–Nb this system serves as a prototype to study SM and SE in Ni-free Ti-based alloys. Its importance is underlined by the fact that most of the recently developed low-modulus as well as SM and SE β-stabilized Ti-based alloys are modifications of the Ti–Nb. Hence, a better understanding of the phase transformations occurred in this binary alloy system will help explain, at least to some extent, the alloys derived from it.
In a cooperation work with partners from BioTiNet EU project (www.biotinet.eu) the mechanical and functional properties of metastable Ti-Nb alloys are optimized by controlled adjustment of the microstructural parameters via complex thermomechanical processing paths.
M. Bönisch et al., Routes to control diffusive pathways and thermal expansion in Ti-alloys, Sci. Rep. 10 (2020) 3045/1-9
M. Bönisch et al., Giant thermal expansion and a-precipitation pathways in Ti-alloys, Nat. Commun. 8 (2017) 1429/1-9
M. Bönisch, et al., Factors influencing the elastic moduli, reversible strains and hysteresis loops in martensitic Ti-Nb alloys, Mater. Sci. Eng. C 48 (2015), 511
M. Bönisch, et al., Thermal stability and phase transformations of martensitic Ti-Nb alloys, Sci. Technol. Adv. Mater. 14 (2013) 55004/1-9
Project: EU ITN BioTiNet
Titanium and its alloys are widely used in medical device industry due to their excellent corrosion resistance, low density, superior mechanical properties and high biocompatibility. A major problem is the reduced strength and hardness of these alloys, which may cause implant fracture. Limited wear resistance can also create the incidence of allergy and inflammatory cascades because of the release of particles and/or toxic metallic ions. Improvement of their mechanical properties plays an important role in enhancing the biomechanical compatibility of metallic implants, leading to a long-term safety and durability in the human body.
The development of composite-type structures based on titanium, can significantly improve the wear resistance, hardness and mechanical strength of implant materials. Our studies are focused on two types of Ti-based composites obtained with different processing routes: a) Ti-matrix composites reinforced with TiB particles, processed by additive manufacturing (selective laser melting-SLM) and powder metallurgy, and b) Ti-based metallic glass-matrix composite, produced by controlled casting.
We highlighted the potential to develop additive manufacturing of novel, low-cost bulk and porous titanium-matrix composites (Ti-TiB) to meet the needs for biomedical orthopaedic implants. We provided a pathway to their development through the application of alloy composition design and reinforcement strategies, manufacturing optimisation and identification of process-structure-property relationships controlling performance in combination with computer-aided structural design tools.
H. Attar et al., Nanoindentation and wear properties of Ti and Ti-TiB composite materials produced by selective laser melting, Mater. Sci. Eng. A 688 (2017) 20
L.-C. Zhang, H. Attar, M. Calin, J. Eckert, Review on manufacture by selective laser melting and properties of titanium based materials for biomedical applications, Materials Technology 31 (2016) 66-76
H. Attar et al., Mechanical behavior of porous commercially pure Ti and Ti-TiB composite materials manufactured by selective laser melting, Mater. Sci. Eng. A 625 (2015) 350
H. Attar et al., Selective laser melting of in situ titanium–titanium boride composites: Processing, microstructure and mechanical properties, Acta Mater. 76 (2014) 13
H. Attar et al., Comparative study of microstructures and mechanical properties of in situ Ti–TiB composites produced by selective laser melting, powder metallurgy, and casting technologies, J. Mater. Res. 29 (2014) 1941

Besides Mg- and Zn-based alloys, Fe-based alloys belong to the new class of biodegradable (bioabsorbable or bioresorbable) metallic materials for biomedical applications. After providing a temporary metal-like functionality they should safely degrade in the human body without the generation of toxic residues.
● Fe-Mn layers were prepared by electrodeposition and by magnetron co-sputtering with envisioned application as component of biodegradable electronic devices and as alternative to polymers or pure metals used as packaging layers. Morphological, chemical, structural and magnetic characterization of the deposited layers was conducted and the biocompatibility characteristics were assessed. By electrodeposition, different ratios of Mn:Fe ions in the water-based sulfate electrolyte led to metallic and oxide based Fe-Mn layers. The electrodeposited layers showed different crystalline structure depending on their nature: a bcc-Fe crystal structure when they were metallic and fcc when they were oxidized. In addition, glycine was used as complexing agent in the electrolytic bath to reduce crack formation and to better control the Mn content of the layers (9-24 wt.%). In comparison, thin Fe-Mn films with Mn contents of 10-70 wt.% which were obtained by magnetron co-sputtering revealed a well-defined bcc structure that lowers its symmetry when the Mn content increases. The corrosion behavior of the deposited layers was evaluated in NaCl solutions by means of electrochemical studies and a trend of reactivity enhancement with increasing Mn content was detected. First in vitro cell tests revealed in particular for the magnetron co-sputtered films a high biocompatible characteristics with a cell viability of 85% even for the Mn-richest sputtered films.
M. Fernandez-Barcia, S. Kurdi et al., Comparative study of the sustainable preparation of FeMn thin films via electrodeposition and magnetron co-sputtering, Surf. Coat. Technol. 375 (2019) 182.
M. Fernandez-Barcia et al., Electrodeposition of mangenese layers from sustainble sulfate based electrolytes, Surf. Coat. Technol. 334 (2019) 261.
Project: EU ITN SELECTA
● Austenitic Fe-30Mn-1C- based alloys are fabricated at the IKM by means of casting and selective laser melting and they are developed with envisioned application as new materials, e.g. for biodegradable stents. For cast Fe-30Mn-1C samples fundamental electrochemical studies were conducted in different NaCl solutions as as well as in simulated body fluids in order to assess their corrosion behaviour. The alloy exhibited in all test solutions – from complex Tris-buffered SBF to non-buffered NaCl solutions with either very low concentration or strong acidification – an actively dissolving nature. This is different to the behaviour of 316L stainless steel used as reference material with a strong passivation tendency. At low NaCl concentrations, an effect of Mn in yielding much higher corrosion current densities of Fe-30Mn-1C compared to passivating pure Fe is detectable. Moreover, local Mn enrichment in austenite grain boundaries of Fe-30Mn-1C causes non-homogeneous surface dissolution in the initial corrosion stage and thus, formation of corrosion patterns. The role of Tris as organic buffer for simulated body fluids is critically assessed in particular with view to the sensitivity of the electrochemical response of Fe. This study supports the need for definition of suitable test conditions for the new class of biodegradable metals. Further studies employing electrochemical measurements and immersion testing with metal release analysis highlighted the positive impact of small S- or B-additions in enhancing the alloy degradations rate while not affecting the cytocompatibility.
M. Otto, V. Shtefan, A. Gebert et al., Mechanisms of laminar flow controlled in vitro corrosion of a biodegradable Fe-Mn-C steel, Corros. Sci. 249 (2025) 112860.
A. Gebert, …, J. Hufenbach et al., Corrosion studies on Fe-30Mn-1C alloy in chloride-containing solutions with view to biomedical application, Materials and Corrosion 69 (2018) 167.
J. Hufenbach, …, A. Gebert et al., S and B microalloying of biodegradable Fe-30Mn-1C - Effects on microstructure, tensile properties, in vitro degradation and cytotoxicity, Mater. Design 142 (2018) 22.
J. Hufenbach, J. Sander, …, A, Gebert et al., Effect of selective laser melting on microstructure, mechanical, and corrosion properties of biodegradable FeMnCS for implant applications, Adv. Eng. Mater. (2020) 2000182


BIOREMIA ("BIOfilm-REsistant Materials for hard tissue Implant Applications") is a 4-year project funded by the European Commission under Horizon 2020 Marie Skłodowska Curie Actions (European Network Training, GA No. 861046).
The key idea behind this project is to address the implant/biomaterial-associated infections problem by developing novel biomaterials and coatings that can reduce both bacterial adhesion and biofilm formation.
Implant-associated infections, commonly caused by biofilm-forming bacteria, are highly problematic for the patient, healthcare system, and society. These biofilms are difficult to treat with conventional antibiotic treatments and novel strategies for avoiding bacterial colonization and biofilm formation is needed.
BIOREMIA aims to tackle this problem by proposing innovative material-based solutions with enhanced antibacterial and antifouling functionality that will result in improved biological acceptance of implants for bone-related applications (orthopedics and dentistry). BIOREMIA scientists will use state of the art materials and surface modification technologies designed to potentially limit the initial stages of bacterial adhesion, as well as biofilm formation.
BIOREMIA research program will be implemented by 15 Early Stage Researchers (ESRs) / PhD students spread across 11 academic and industrial institutions from 10 European countries (Germany, Austria, Italy, Sweden, Greece, UK, Spain, Ireland, France, and Switzerland). Two BIOREMIA PhD students are hosted by our department:
Background information on all ESR projects and BIOREMIA Network activities is available on www.bioremia.eu
Contact:
Assoc. Prof. Dr. Mariana Calin (BIOREMIA coordinator)
Phone: +49 351 4659 613
E-mail: m.calin(at)ifw-dresden.de