
One of the most important thin film deposition techniques conducted in our group is (m
etalo
rganic) c
hemical v
apor d
eposition, short (MO)CVD. Thin films are grown from volatile metal complexes from the gas phase. Using a carrier gas flow, the evaporated the precursor is transported to a heated substrate, where a diffusion through a thin boundary layer can lead to an adsorption of the precursor on the substrate surface. From here, different events can happen: The precursor can either decompose due to high substrate temperatures or low thermal stability of the complex itself or it can react with a co-reactant on the surface forming small islands of material ultimately resulting in the growth of the thin film. Notably, the precursor molecules are non-stationary and can diffuse along the substrate surface before reacting. After the reaction/decomposition the by-products ideally desorb from the surface of the substrate.
CVD is a rather complex thin film process due to its continuous operation mode: Process characterisics such as flow dynamics, kinetics, reactivities and side-reactions need to be well-balanced when designing CVD processes.
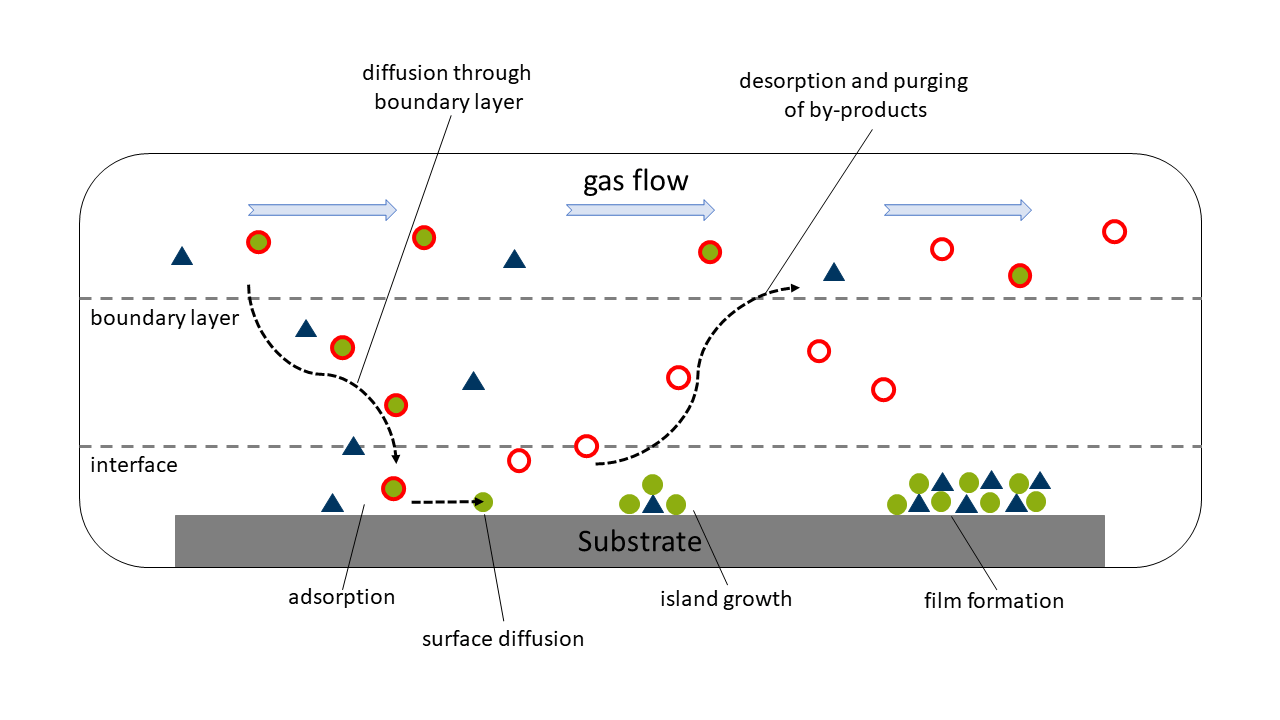 Schematic of the typical steps of a CVD process.
Schematic of the typical steps of a CVD process.A prototypical CVD process is conducted under reduced pressure in a substrate temperature range of ~300 °C up to 1000 °C. The thickness of thin films produced via CVD usually range from few nm to several µm range.
